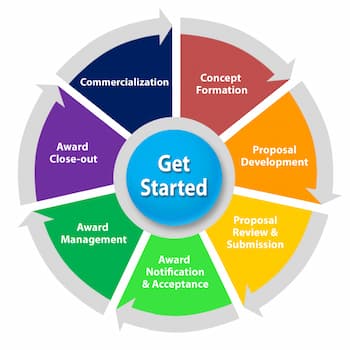
Proposal development is a team effort and can benefit from the early involvement of the Grants and Contracts Administrator (GCA), potential collaborators, and guidance from a mentor or seasoned investigator.
To get started, the Principal Investigator (PI) will need to:
- Identify a research idea, funding opportunity and confirm eligibility
- Review the funding announcement (with the GCA)
- Establish a timeline
- Create proposal components (research strategy, budget, etc.)
Duke uses Sponsored Projects System (SPS) to prepare, route and manage all proposal submissions and awards. For each proposal, an SPS record is created by the GCA and routed for institutional review. Before the SPS record can be generated, the PI needs to provide the following:
- Funding Announcement
- Budget
- Budget Justification
- Project Title
- Draft Abstract
- Protocol Information (IRB, IACUC, IBC)
- Key Personnel List
To ensure the proposal routes smoothly, the PI will need to work with the GCA to ensure all required elements have been addressed.
When in doubt about how to proceed, reach out to the GCA for assistance. Duke is committed to supporting research and the Department of Medicine has access to the many resources at Duke, as well as staff with many years of experience to support this mission.
In order to apply for a grant through Duke University, an individual must hold PI status. The status of PI is granted to all individuals with faculty appointments. For employees that need to acquire PI status for an application, please refer to the Procedure for Requesting Eligibility To Serve as Principal Investigator.
Sponsors communicate the purpose, criteria and guidelines of a funding opportunity through a funding announcement. A funding announcement specifies the:
- Type of funding mechanism (grant, contract, cooperative agreement or gift)
- Sponsor agency
- Application due date
- Maximum project period
- Eligibility criteria and application guidelines
- Conditions or restrictions to consider prior to preparing an application
Eligibility or other sponsor criteria can impact the viability of an application, so it is important to uncover these factors as soon as possible. Include the GCA in the review of the funding announcement to identify application components and non-standard aspects of the announcement.
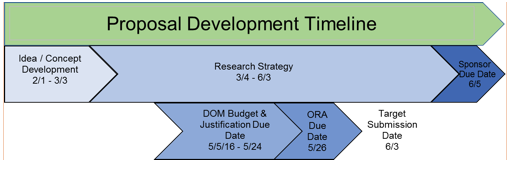
Developing a timeline for the preparation of an application can help the PI successfully meet the internal and sponsor deadlines for a submission. It also allows the GCA and the PI to coordinate efforts in the creation of application materials and ensure consistency throughout each section. It is recommended that the PI:
- Begin the research strategy and budget 2 months prior to sponsor deadline
- Notify the GCA of intent to submit at least 30 days prior to sponsor deadline
- Provide the GCA with final budget and budget justification, as well as other designated administrative sections, 20 days prior to sponsor deadline
- Submit proposal to Office of Research Administration (ORA) for review at least 7 business days before sponsor due date
To develop a timeline start with the sponsor due date and work backwards, as is shown in this example for an R01 submission due June 5.
Regardless of the sponsor, an application typically requires a research strategy and a budget for the proposal. Other proposal sections may be identified in the funding announcement.
Research Strategy
The research strategy typically describes the significance, innovation and approach of the research. The PI’s approach and the experiments that will be done to accomplish the aims should be explained, as well as the context for the research, including preliminary data, literary citations, and anticipated results and implications. For consistency, the research strategy and budget creation should be developed simultaneously.
The Office of Research Development provides services to teams of investigators and individual investigators to help ensure submission of high quality grant applications. Services are available on a first come, first serve basis. They also offer writing workshops.
Budget
A budget outlines the expenses required to successfully complete the proposed research. The amounts listed should be a reasonable estimation of the actual costs and should be consistent with the application narrative. Inconsistencies can put the proposal at risk. It is not recommended to match the budget to the sponsor’s maximum funding level. If the budget total is below the minimum funding level consider additional experiments or sub-aims that would strengthen the proposed research.
As a starting point, the GCA will provide the PI with a proposal tool to identify the application sections where funds are requested. When creating the budget there are six main budget categories:
- Personnel (Salary and Fringe Benefits)
- Supplies
- Patient Care Costs
- Other Expenses
- Equipment
- Indirect Costs
- Subcontractors/Collaborators
Personnel
Personnel costs support the professional effort necessary to satisfactorily complete the goals and aims of the project. Proposed effort should be a reasonable estimation of the time it takes for the identified personnel to complete their roles on the project, and should be determined using the institutional base salary for each individual. This applies to all personnel, from the PI, to collaborators, research technicians, postdoctoral associates and other personnel. If certain personnel are needed, but named individuals are not yet known, the GCA can help determine a salary range based on the anticipated level of experience.
Supplies, Patient Care Costs and Other Expenses
When budgeting for supplies, patient care costs, and other expenses (such as facility user fees) use a reasonable estimation as to what items are needed to complete the proposed research project and the costs associated.
Equipment
Any item that is necessary to complete the work of the proposed research, exceeds $5,000, has a useful life of more than 1 year and is not currently available through Duke is considered equipment and can be requested in a project budget. It is important to note that indirect costs are generally not applied to equipment expenses, which can affect the total budget for the project. When budgeting for equipment, it is best to discuss the needs with your GCA and a faculty mentor (if available), who can determine if it is appropriate to include in the budget.
Indirect Costs
Indirect Costs, also known as Facilities and Administrative (F&A) costs, are expenses incurred for a shared or common objective and cannot readily be associated with a specific project, such as administration. These rates are set by Duke via the negotiated rate established with DHHS. It is necessary to consider these costs when funding announcements restrict the total funding limit for a project. Otherwise, if the funding limit is direct cost based, the indirect costs do not need to be considered when establishing the budget.
Collaborators
When collaborators are necessary to successfully execute the research, the PI should have discussions early on to better understand that relationship. There are three types of external collaborators:
- Subrecipient/Subawardee/Subcontractor: A third-party entity that receives funding from the Duke University to carry out activities related to the project’s scope of work. The subrecipient plays an integral role in the project and is involved in programmatic decision-making.
- Contractor: A dealer, distributor, merchant or other entity (which may include an outside institution or university) that provides goods or services required for the conduct of the project. These goods or services may be for Duke’s own use or for the beneficiaries of the project.
- Consultant: An individual or firm that provides professional advice or services for a fee, but not normally as an employee of the prime organization. In very rare circumstances, an individual may be both a consultant and an employee of the prime organization.
Contact the GCA if unsure which best categorizes the project’s external collaborator. Determining the type of collaborator will help establish a budget for the subcontracted work. For example, indirect costs are charged on a portion of a subcontractor’s expense, but are charged on the full expense of a consultant. The GCA should be notified of subcontractor involvement in the proposed research well in advance of the budget due date. As with all budget requests, the subcontractor’s budget should be based on actual needs to complete the proposed work or services.
Other Proposal Sections
Additional sections of the proposal are identified in the funding announcement and often include:
- Abstract
- Specific Aims
- Responsible Conduct in Research
- Human Subjects Protections with IRB protocols/clinical research
- Vertebrate Animals for research with IACUC protocols/research
- Letters of Support
- Biographical Sketches
Each item is an additional section and requires separate documentation to be attached in the proposal. Communicating with the GCA early in the proposal development process and thorough review of the funding announcement will help to make sure all required sections are completed. The GCA can provide examples of sections, if needed.
Biographical sketches need to be completed in the correct format. Different funding agencies have different formats, specifically DOD, NIH and NASA, even though they are all federal sponsors. NIH biographical sketches can be created using existing Science Experts Network Curriculum Vitae (SciENcv) profiles. For assistance in determining the correct format, contact a GCA.