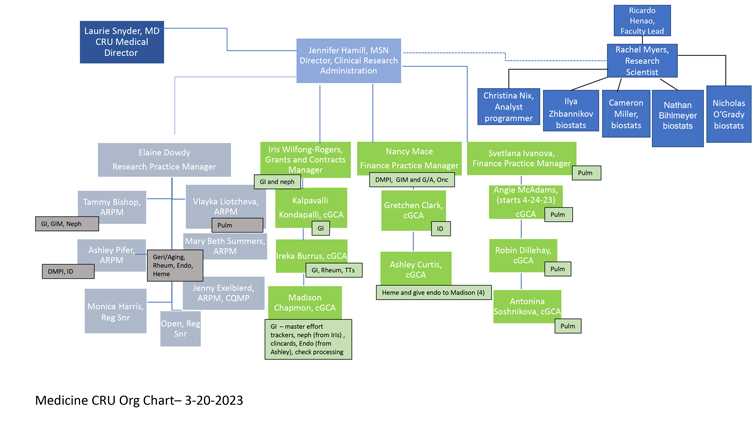
The Chair of the Department of Medicine has the ultimate responsibility and accountability for the oversight of all CRU activities. The Medicine CRU Medical Director is appointed by the Chair and reports directly to the Vice Chair of Research who reports directly to the Chair of the Department of Medicine.
Leadership
The Medical Director has the ultimate responsibility for all research involving human subjects in the Department of Medicine. The Medical Director will provide support of the operational staff in the Medicine CRU, providing oversight for the Director of Clinical Research Administration to whom direct reporting occurs from the Research Practice Manager, Finance Practice Managers, and Director of the Bioinformatics and Clinical Analytics Team (BioCATs).
The Director of Clinical Research Administration is responsible for high level oversight and effective management of the operations and finances of the CRU. In addition to providing oversight to the CRU Operations Team, the Director of Clinical Research Administration will lead innovations and initiatives for improving the CRU user experience and addressing feedback on performance concerns of staff and the overall CRU. This is a departmental administrative leadership position that will focus on improving the function, efficiency, and service provided by the CRU and developing new programs and initiatives that address deficits in the mission.
Operational Staff
The Research Practice Manager (RPM) will manage the day-to-day operations of the CRU related to study conduct including IRB submissions and renewals; protocol initiation; participant recruitment, screening, consent and enrollment; safety reporting; and study close-out, record retention and audit ready files stored. The RPM will oversee the departmental assistant RPMs (ARPMs) to whom the study conduct and regulatory compliance duties of all division Clinical Research Professionals (CRPs) report. This oversight includes, but is not limited to: implementing strategies to support the proposed studies, monitoring regulatory documentation and practices, development of SOPs to standardize research support and training, orientation and ongoing training of CRPs, communication with PIs as well as sponsors or other sites, human resource management of clinical research staff, financial oversite of clinical research studies, quality assurance reviews of study documentation and management, and overall problem solving and management of any issue that arises in the conduct and management of the research.
The Assistant Research Practice Managers (ARPM) is assigned to a single or a number of divisions, which includes institutes or centers, within the Department of Medicine (DOM). The DOM ARPMs will work closely with the CRP who is assigned a lead coordinator role (ie., RPL, RPL Snr, CRC, or CRNC Snr) in each division while providing upper level management across the entire study team to ensure accountability for timely project progression and metrics are met as well as adherence to regulatory compliance. One ARPM is devoted to the oversight of Clinical Quality Management (CQMP) which is designed to provide oversight of studies that are not otherwise externally monitored. More information here: https://dosi.duke.edu/quality-management-programs/clinical-quality-management-program-cqmp
The Regulatory Coordinator Senior (RC, Snr) is responsible for the regulatory oversight for the DOM. The RC Snr ensures all faculty and key personnel on Medicine CRU protocols are current on all applicable research training and facilitate the review process on all new protocols submitted to the IRB. The RC Snr also coordinates physician faculty attendance at 96 IRB meetings each year.
The Finance Practice Manager (FPM) is responsible for ensuring the financial feasibility for all clinical research within the DOM Department of Medicine, including all pre- and post-award activities. The FPM should have an understanding of the non-financial aspects of the overall process of research projects, including but not limited to billing and regulatory, understand the contract review and SPS entry processes and provide approval of budget and payment terms to ORC. The FPM should understand reconciliation of patient care costs and non-patient care costs, follow and implement effort management and reporting policies, understand and monitor revenue management and understand programmatic codes and SAP reports for financial management. The FPM should become knowledgeable of the portfolio of studies being reported on, sharing applicable information with investigators, CRU, Chair and/or Business managers. The FPM understands the Clinical Research Closeout Policy, processes/reviews/approves the Closeout Tasklist and maintains/destroys accounting documents associated with study-specific cost objects. The FPM understands what a Hub Code is and what costs are managed within them. The FPM collaborates with the Grants Management Team to ensure they understand the processes of the Institution and the School of Medicine regarding pricing, budgeting, effort management and patient care charged to grants, ensuring that they have processes in place to manage non-industry clinical research projects and have access to the necessary applications. The FPM ensures the Grants Management Team understands Maestro Care reporting and reconciliation, assists them with resolving billing and revenue issues and maintains joint collaborations between the CRU and Grants Management Team. The FPM should be able to work fluently in the OnCore Clinical Trials management system which records and tracks finances related to study activity and which creates invoices and in which the FPM will reconcile payments from sponsors. The FPM will oversee a team of clinical Grants and Contracts Administrators (cGCAs) to ensure proper oversight of all financial responsibilities for which they are responsible. The FPM will track SOM metrics and make efforts to meet those metrics.
The Bioinformatics and Clinical Analytics Team (BioCATs) joined the Medicine CRU in July 2022. Please use this link to submit a request for stats support and to view their scope of work https://medicine.duke.edu/research/research-resources/bioinformatics-and-clinical-analytics-team